How will CRISPR gene editing change healthcare?

For years, one medical technology has captivated the scientific community: CRISPR.
In 2012, Emmanuelle Charpentier, a microbiologist, geneticist and biochemist, and Jennifer A. Doudna, a biochemist, published a paper detailing their contributions to the development of the CRISPR-Cas9 gene editing tool, eventually winning the 2020 Nobel Prize in Chemistry for their teams work.
CRISPR is a type of gene editing, also known as genome editing. According to the National Library of Medicine, this refers to the practice of changing a living thing’s DNA, and may involve changing, removing or adding in genetic material. And it has incredible potential.
“This technology has had a revolutionary impact on the life sciences, is contributing to new cancer therapies and may make the dream of curing inherited diseases come true,” the Nobel Prize press release for Doudna and Charpentier announced.
What is CRISPR? How does it work?
When humans develop a viral infection, the immune system produces antibodies against the virus so it can respond quickly the next time it is threatened.
Bacteria have a similar response, known as Clustered Regularly Interspaced Short Palindromic Repeats and CRISPR-associated protein 9. It’s commonly called CRISPR-Cas9 or just CRISPR.
When infected, many bacteria gather pieces of the viral code and tuck them into their own genomes for safekeeping. If that same virus shows up again, the bacteria recognizes it and unleashes a DNA-cutting protein called Cas9 to chop up the invader’s genetic code.
CRISPR is “a bacterial acquired immune system, essentially. So this is a defense mechanism that bacteria evolved millions of years ago, to survive attacks from viruses,” says Ryan T. Wagner, Ph.D., a researcher of epigenetic etiology of human disease at Mayo Clinic.
Researchers observed this biological process and, over decades of research, engineered a way to develop a CRISPER/Cas9 gene-targeting system that’s compatible with the human genome.
“We’ve taken this adaptation from nature and applied it as a technology to perform, essentially, gene targeting,” says Dr. Wagner.
CRISPR enables researchers to cut and paste DNA sequences. First, scientists compose a string of genetic letters or “guide RNA” that, like those original snippets of viral code, can recognize a specific stretch of DNA in the genome. Second, they introduce this guide sequence to the target cell, along with an enzyme like Cas9, which recognizes the matching text and cuts it open. Scientists use this mechanism to delete, mutate, insert or repair genomic DNA sequences in cells, animals and humans.
“We can use these biological elements such as RNA to direct proteins to specific sites in the genome, and that was really transformative,” says Dr. Wagner.
When CRISPR-Cas9 came on the scene, another gene editing technology called TALEN already existed. While it is still in use, CRISPR has become the more popular option — something Dr. Wagner has seen firsthand in the research space.
How can CRISPR gene editing transform healthcare?
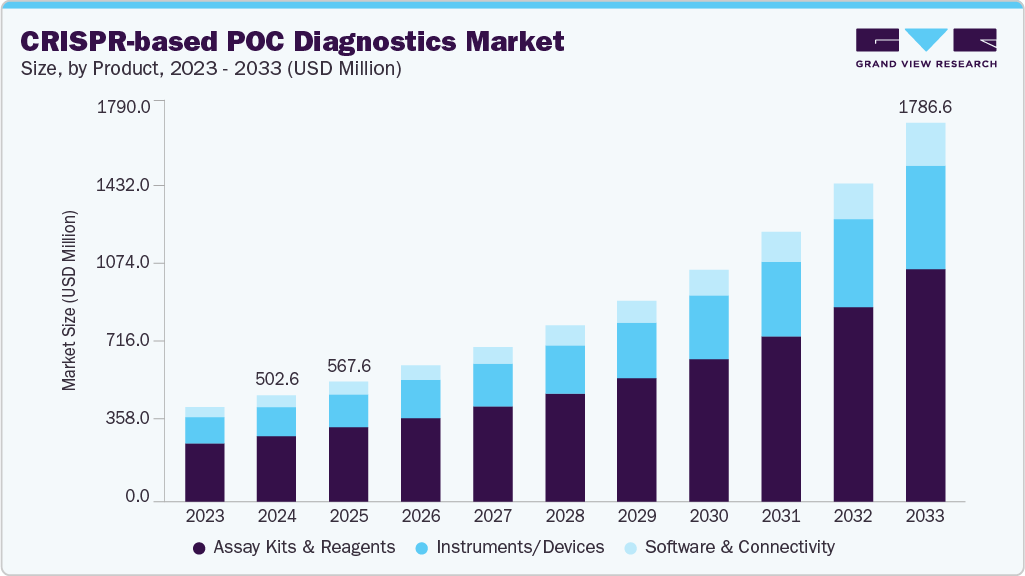
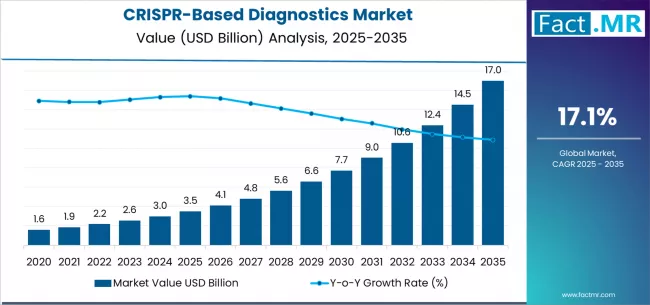
A great deal of the work involving CRISPR has taken place in research and clinical settings. Dr. Wagner notes that CRISPR has already been transformative in how he conducts research, but that the average person has yet to see its impact in their daily lives. But he believes that will change soon, as CRISPR is used to create diagnostic tests.
As a result of the COVID-19 pandemic, many people became familiar with rapid diagnostic testing. Unlike the rapid tests for COVID-19, which detect certain proteins in the virus, CRISPR recognizes specific nucleotide sequences at the DNA or RNA level, which Dr. Wagner says allows for greater sensitivity.
“We’ve become so accustomed to testing at home in the past four years, so I think people will start to expect that more and more and be more comfortable with it,” he says.
CRISPR diagnostic tests are being developed to identify primarily bacterial infections, especially in the agricultural space with livestock. Dr. Wagner believes these tests will quickly move into a more clinical space, and soon become relatively commonplace.
Individuals could conduct these at-home tests to give their healthcare professionals more information — ideally allowing a healthcare team to quickly detect certain conditions and provide better treatment. Dr. Wagner adds that providing this data from home could save people and medical professionals money, providing healthcare professionals with greater insight before a patient even steps into their office.
While the technology exists to detect both bacteria and viruses, Dr. Wagner sees most molecular diagnostic tests as being in the bacterial space initially. A key difficulty with any of these tests is manufacturing a low-cost yet adequately sensitive test that has a good shelf life and doesn’t require other instruments to run on.
CRISPR also shows promise as a way to treat or cure genetic diseases by editing DNA mutations associated with the condition.
Looking forward with CRISPR
Dr. Wagner predicts that in the next 10 years, there will be a rise in therapeutic applications for CRISPR, including in CAR-T therapy and cancer treatment, and that these could be available even outside of clinical trials.
Healthcare is already moving in that direction: In December 2023, the Food and Drug Administration approved Casgevy, the first gene therapy for treating sickle cell disease which utilizes CRISPR. In addition, many successful clinical trials using gene therapy have been completed. As one example, a recent trial reported on successful treatment of Leber congenital amaurosis, an inherited form of blindness.
Dr. Wagner notes that CRISPR technology continues to advance in both the clinical and the therapeutic spaces, but it must move slowly due to the high stakes involved.
“You don’t want to create mutations while you’re repairing the one mutation you’re after,” he says. “And that’s the real risk of using CRISPR. Doing these treatments inside you makes it harder to mitigate those risks.”
Part of this risk stems from the fact that these treatments can cause nonspecific activity — activity outside where it’s being directed — within the body.
“I certainly know from the research side that when you deliver CRISPR-cas9 … that it can have activity outside of where you’re directing it.”
He believes there are ways that clinicians can mitigate these risks.
He sees new research papers coming out regularly about potential modifications or applications of CRISPR and believes it will continue until the next “transformative discovery” in this field.

Relevant reading
Mayo Clinic on Hearing and Balance, 3rd Edition
Mayo Clinic on Better Hearing and Balance helps readers understand the possible causes of hearing and balance issues and offers solutions aimed at improving not just hearing and balance, but quality of life overall.
link